This is due to the high prevalence of pathologies such as osteoarthritis (OA), which affects approximately 27 million adults in the United States and can drastically reduce quality of life. In the field of biomechanics, a strong motivation to better understand the mechanics of articular cartilage exists. For this reason, the search for a better understanding of multi-scale spatial interactions has become an increasingly desirable objective, in order to establish the causal mechanical relationships between the loading of joints, tissues, and cells. While cellular activity at the micro-scale alters the mechanical environment, loading transferred from higher spatial scales also plays a role. Often, research has been conducted at the spatial scale of the cell and its immediate extracellular environment. The phenomenon of cell behavior being directed by mechanical stimuli, referred to as mechanotransduction or mechanoregulation, as well as cell damage resulting from mechanical disruption, have been a topic of research in medicine and biology for several decades –. The method’s generalized nature also allows for substitution of any macro-scale and/or micro-scale model providing application for other multi-scale continuum mechanics problems. The coupling method proved to be highly scalable due to micro-scale model independence that allowed for exploitation of distributed memory computing architecture. In the 11 cell case, all cells experienced less deformation than the single cell case, and also exhibited a larger variance in deformation compared to other cells residing in the same block. In both cases, chondrocytes experienced amplified deformations compared to those at the macro-scale, predicted by simulating one body weight compressive loading on the tibiofemoral joint. To illustrate further utility of this multi-scale modeling pipeline, two micro-scale cartilage constructs were considered: an idealized single cell at the centroid of a 100×100×100 μm block commonly used in past research studies, and an anatomically based (11 cell model of the same volume) representation of the middle zone of tibiofemoral cartilage. Various cellular deformation metrics (aspect ratio change, volumetric strain, cellular effective strain and maximum shear strain) were calculated. It was possible to identify the load distribution within the knee among its tissue structures and ultimately within the cartilage among its extracellular matrix, pericellular environment and resident chondrocytes. In this study, a multi-scale analysis pipeline was implemented to post-process the results of a macro-scale finite element (FE) tibiofemoral joint model to provide joint mechanics based displacement boundary conditions to micro-scale cellular FE models of the cartilage, for the purpose of characterizing chondrocyte deformations in relation to tibiofemoral joint loading. in the cartilage, is not a straightforward task.
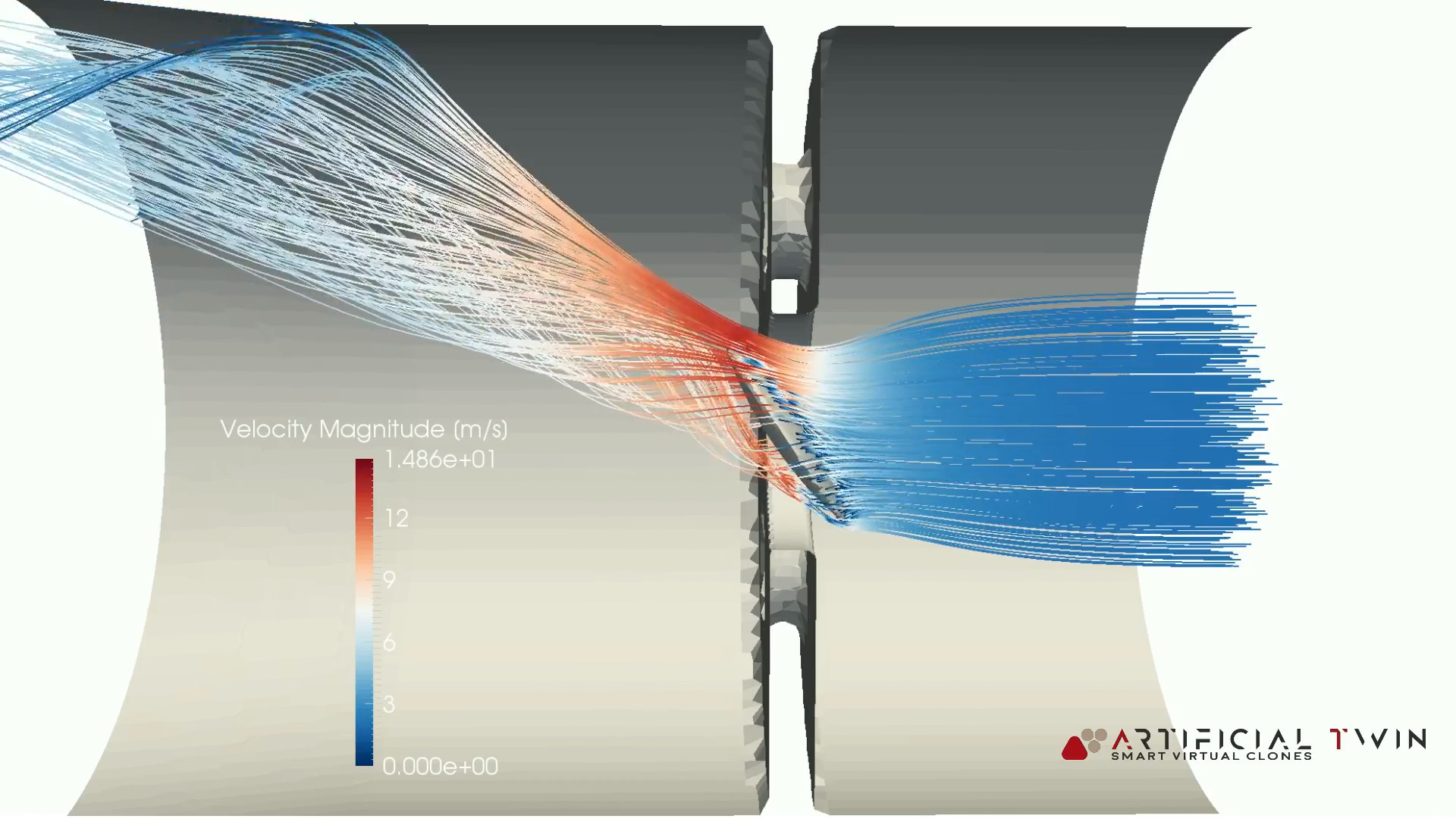
However, understanding how joint level loads relate to cell level deformations, e.g. Cells of the musculoskeletal system are known to respond to mechanical loading and chondrocytes within the cartilage are not an exception.